|
When knowing this, biomarkers can be categorized into different classes depending on the information they provide. Here are some examples of what they can mean in pain research: a biomarker can distinguish different underlying pain mechanisms (= diagnostic biomarker) or can reveal which patients have a higher chance to develop (chronic) pain (= susceptibility biomarker) or can predict which patients will develop chronic pain after (breast) cancer treatment (= prognostic biomarker) or can anticipate which patients will benefit from pain education (= predictive biomarker) [2, 3]. Since biomarkers have different purposes in practice, they are useful to gain more knowledge on different levels of pain. Eventually, both clinicians and patients can take advantage of them: clinicians are more aware of the patients’ pain experience and thus can provide better healthcare, while today chronic pain remains underrecognized and undertreated subsequently negatively affecting the patient’s quality of life.[4, 5].
Up to now, no biomarkers are identified for chronic pain [6]. However, a lot of potential biomarkers are taken under the loop to better differentiate between pain pathologies, predict patients' responses to personalized treatment and improve the quality of life. The biggest hurdle to finding biomarkers for (chronic) pain is that pain can’t be objectively measured. This is not surprising since pain is defined by the International Association for the Study of Pain as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.” This indicates the perception that the experience of pain can be different in each individual. Today, the golden standard to measure pain remains with the usage of (subjective) self-reports (see Figure). However, a combination with more objective assessments or measurable biomarkers would be useful to better understand the underlying mechanism of pain and to improve personalized pain management [3]
Figure: Possibilities to measure (chronic) pain. QST = quantitative sensory testing
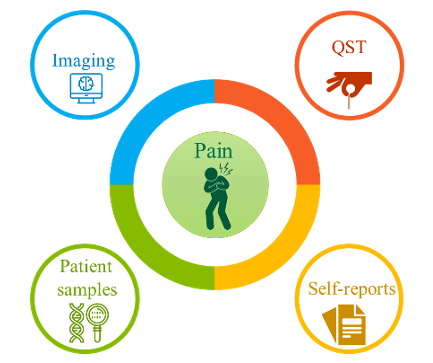
A more possible objective assessment came with the appearance of quantitative sensory testing (QST). QST can contribute to a better diagnosis of the mechanisms of (chronic)pain in combination with self-reports. This commonly used technique gives standardized stimuli to the patients, whereas the outcome measurement is still influenced by the patient’s sensory experience. Since there is a subjective note to QST and thus not consistent with the BEST definition, it is under debate whether it could serve as a (potential) biomarker. However, we can’t throw the baby out with the bathwater. QST remains an interesting technique to study pain: besides the diagnostic use, it also can have a prognostic and predictive value. It can be used to indicate pain progression or severity but also treatment response. Nevertheless, further research is necessary to validate the applied QST method and to study its reliability [6]
What are interesting potential biomarkers for pain?
Currently, the interest in the pain research field to find potential biomarkers for chronic pain has increased, in particular in the domain of neuro-imaging and (epi)genetics [3, 7].
Neuro-imaging is considered to most promising biomarker in the field of pain. Depending on the acquisition, it can be used to look at anatomical changes, connectivity changes or neurochemical alterations [3]. Evidence shows reduced gray matter volume and abnormalities in white matter and brain connectivity in chronic pain patients (=diagnostic) [6]. Besides, common comorbidities of chronic pain could be imaged such as anxiety, depression and catastrophizing. This could give an added and more objective value to the patients’ self-reports [3, 6]. The biggest challenge of neuroimaging lies with delivering personalized information instead of comparing groups, which could give a prognostic or even predictive value [6].
Next to neuroimaging, a shift is made in the field of genetics: a long time is searched for alterations in one gene causing pain. Until a certain moment, researchers found polygenic alterations underlying a specific condition. For example; the risk of developing breast cancer by mutations in the BRCA1&2 genes [3]. This change of view is interesting since (chronic) pain has no single cause and could lead to the basis in order to distinguish different pain phenotypes including post-surgical, musculoskeletal and neuropathic pain [6]. Besides, more recent evidence suggests a relation between the central nervous system and the immune system, which need to be further exploited [8, 9]. For this purpose different samples such as blood, urinary, saliva and cerebrospinal fluid are taken under the loop. Not only looking at fundamental genes to identify possible risks to develop (chronic) pain but also proteomics, transcriptomics, metabolomics and epigenetics are rising features to see the whole pain picture [8].
The area of investigating pain biomarkers seems very promising and gives bright expectations. However, some limitations need to be taken into account: the timing of biomarker analysis, correcting for comorbidities and consent to study patients are very common challenges [7]. The research of finding pain biomarkers comes with accompanying hurdles but the last part of this chapter is definitely not written yet. As in any research, there is always a catch.
Drs. Amber De Groote
References
1. Aronson, J.K. and R.E. Ferner, Biomarkers-A General Review. Curr Protoc Pharmacol, 2017. 76: p. 9 23 1-9 23 17.
2. Group., F.-N.B.W. 2016.
3. Tracey, I., C.J. Woolf, and N.A. Andrews, Composite Pain Biomarker Signatures for Objective Assessment and Effective Treatment. Neuron, 2019. 101(5): p. 783-800.
4. Greco, M.T., et al., Quality of cancer pain management: an update of a systematic review of undertreatment of patients with cancer. J Clin Oncol, 2014. 32(36): p. 4149-54.
5. Pachman, D.R., et al., Troublesome symptoms in cancer survivors: fatigue, insomnia, neuropathy, and pain. J Clin Oncol, 2012. 30(30): p. 3687-96.
6. Smith, S.M., et al., The Potential Role of Sensory Testing, Skin Biopsy, and Functional Brain Imaging as Biomarkers in Chronic Pain Clinical Trials: IMMPACT Considerations. J Pain, 2017. 18(7): p. 757-777.
7. Sisignano, M., et al., Potential biomarkers for persistent and neuropathic pain therapy. Pharmacol Ther, 2019. 199: p. 16-29.
8. Garcia-Gutierrez, M.S., et al., Biomarkers in Psychiatry: Concept, Definition, Types and Relevance to the Clinical Reality. Front Psychiatry, 2020. 11: p. 432.
9. Vanderwall, A.G. and E.D. Milligan, Cytokines in Pain: Harnessing Endogenous Anti-Inflammatory Signaling for Improved Pain Management. Front Immunol, 2019. 10: p. 3009.
10. Hagedorn, J.M., et al., How Well Do Current Laboratory Biomarkers Inform Clinical Decision-Making in Chronic Pain Management?Journal of pain research, 2021. 14: p. 3695-3710.
Pictograms by the Noun Project.
|